
Hospitals
Supply continuity, lot traceability, and product review packets for multi-department deployment.

Chattanooga devices are engineered for the workflow of each care environment, from ambulatory clinics to hospital-at-home monitoring programs.
Clinical application planning starts with the environment, not the product catalog. A dental clinic thinks in operatories, sterilization flow, patient turnover, and infection prevention. A remote care program thinks in patient onboarding, device adherence, data review, and escalation. A supply chain director thinks in lot control, contract pricing, substitution rules, and recall response. Chattanooga structures application guidance around those practical differences so each care setting can evaluate the same product family through its own operational lens.
This page groups Chattanooga programs by care setting to help clinical teams request the right documentation. The same device may appear in more than one environment, but its implementation notes can change. A pulse oximeter used in a clinic may need a quick cleanability and calibration file; the same class used in remote care may need patient instructions, connectivity review, and support escalation. Consumables used in an ambulatory surgical center may need kit logic and sterilization packaging records; similar products in a dental practice may need chairside storage and procedure turnover guidance.

Supply continuity, lot traceability, and product review packets for multi-department deployment.

Procedure-ready kits, fast reordering, and compact documentation for outpatient workflows.
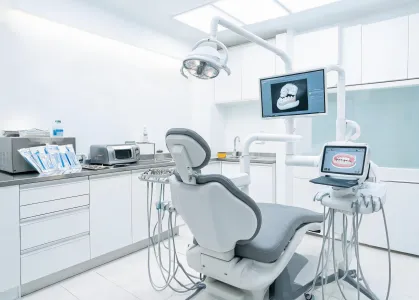
Operatory equipment, scanner workflows, implant planning, and infection control support.

Patient onboarding, connected device support, and review pathways for monitoring data.

Focused configurations for cardiology, wound care, oral health, and rehabilitation service lines.

Caregiver education, device replacement flows, and support routing for distributed programs.